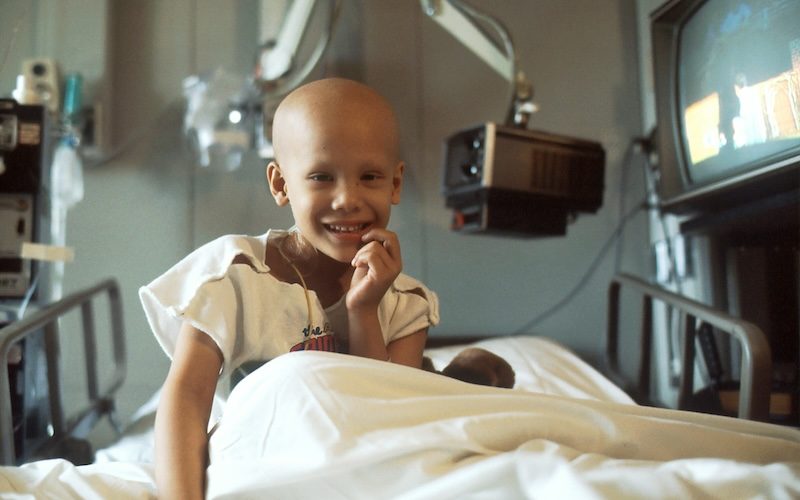
The first at-home brain cancer treatment for children and young people with brain tumours will be made available by the NHS in the coming months.
The NHS will provide children and young people aged one to 17 with low-grade gliomas that have a BRAF V600E genetic mutation with a combination treatment of Dabrafenib with trametinib, which clinical trials have shown slows the progression of the disease.
Approved by the National Institute for Health Care and Excellence (NICE), the at-home self-administered treatment is revolutionizing the world of cancer treatment, as it allows children to be treated in the comforts of their own homes and avoid spending time in the hospital.
Director of medicines evaluation at NICE, Helen Knight, said: “I think this is just such a fantastic recommendation for an incredibly needy population.
“The impact that this disease has on people and their families, we’ve got something that is much more tolerable for patients versus chemotherapy, much easier for all involved, and life-extending as well.”
She added: “We’ve aimed to produce this guidance as quickly as we can, so people can access them as quickly as possible.”
NICE is an organisation that evaluates new medications, treatments, and health technologies for the NHS and approximates their value and clinical effectiveness.
Approximately 150 children are diagnosed with low-grade gliomas and 30 with high-grade gliomas in the UK every year, according to the NHS.
Gliomas are tumours that grow in the brain or spinal cord that depending on growth rate and size, can be deadly.
Knight said the medication is given in the form of dissolvable tablets, to make it easy for children to take and for adults to administer at home without difficulty.
Director of External Affairs and Strategy at the Brain Tumour Charity Cameron Miller said: “It’s really exciting.
“The clinical trial results show potentially delaying tumor growth for more than two, just over two years on average. As well as fewer side effects, the ability to take it home, which means it places less burden, not only on the NHS, but also on the families who are already coping with this diagnosis and so much sort of administration that goes alongside it.”
He added: “It’s heartwarming and joyous that we have this new treatment, but more needs to be done.”
Director of Services and policy at the charity Braintrust Dr Helen Bulbeck spoke of the organisation’s work supporting people with brain cancer and their families as they come to terms with their diagnoses and treatment plan.
Speaking on how they will work with families using the new treatment, Bulbeck said: “I think this is about building people’s confidence so they’re able to use it in their own home.
“Also, building peer-to-peer networks, a growing community of other families that are in the same space so people can share their experiences, because there’s nothing like connecting with somebody else who’s in the same space that you are.”
Bulbeck said: “I think it’s really important to realize that NICE is actually on the patient side.”
She added: “I just wish it would benefit more than 30 children.”
This new brain cancer treatment will allow a small cohort of children and young people to begin their healing process from home without experiencing the harsh side effects of chemotherapy.